Scripps Research Institute: Tacrolimus and astemizole may be effective in treating human prion disease
Prion diseases or transmissible spongiform encephalopathies (TSEs) are a family of rare progressive neurodegenerative disorders that affect both humans and animals, according to the CDC. Contracting this disease attributed to an abnormally folded protein is an automatic death sentence.
However, scientists from the Florida campus of The Scripps Research Institute (TSRI) have for the first time identified a pair of drugs already approved for human use that show anti-prion activity and, for one of them, great promise in treating these universally fatal disorders, according to a Scripps news release today.
According to the study, “Unique drug screening approach for prion diseases identifies tacrolimus and astemizole as antiprion agents”, published in the journal Proceedings of the National Academy of Sciences. The study was led by TSRI Professor Corinne Lasmézas and performed in collaboration with TSRI Professor Emeritus Charles Weissmann and Director of Lead Identification Peter Hodder.
According to the study abstract:
Because prion protein (PrP) is necessary for prion replication but dispensable for the host, we developed the PrP–FRET-enabled high throughput assay (PrP–FEHTA) to screen for compounds that decrease PrP expression.
They screened a collection of drugs approved for human use and identified astemizole and tacrolimus, which reduced cell-surface PrP and inhibited prion replication in neuroblastoma cells.
Tacrolimus reduced total cellular PrP levels by a nontranscriptional mechanism. Astemizole stimulated autophagy, a hitherto unreported mode of action for this pharmacophore. Astemizole, but not tacrolimus, prolonged the survival time of prion-infected mice. Astemizole is used in humans to treat seasonal allergic rhinitis in a chronic setting.
Given the absence of any treatment option for CJD patients and the favorable drug characteristics of astemizole, including its ability to cross the blood–brain barrier, it may be considered as therapy for CJD patients and for prophylactic use in familial prion diseases. Importantly, our results validate PrP-FEHTA as a method to identify antiprion compounds and, more generally, FEHTA as a unique drug discovery platform.
Lasmézas noted that astemizole appears to stimulate autophagy, the process by which cells eliminate unwanted components. “Autophagy is involved in several protein misfolding neurodegenerative diseases such as Alzheimer’s, Parkinson’s and Huntington’s diseases,” she said. “So future studies on the mode of action of astemizole may uncover potentially new therapeutic targets for prion diseases and similar disorders.”
The prion diseases or TSE are characterized by progressive, deterioration of the brain. The human prion diseases are always fatal.
The TSE’s include Creutzfeldt-Jakob disease and all its variants, kuru and Gerstmann-Staussler-Scheinker syndrome.
Though not fully characterized, a prion is an abnormally folded protein which can have long incubation times of many years.
Human prion disease can be acquired through diet, medical treatment, surgery or blood transfusion and some are considered genetic. There is currently no treatment for human prion disease.
For more infectious disease news and information, visit and “like” the Infectious Disease News Facebook page
Looking for a job in health care? Check here to see what’s available
Giardia in Black and White: T-shirts, coffee mugs and other merchandise with a clean, computerized image of a Giardia intestinalis trophozoite
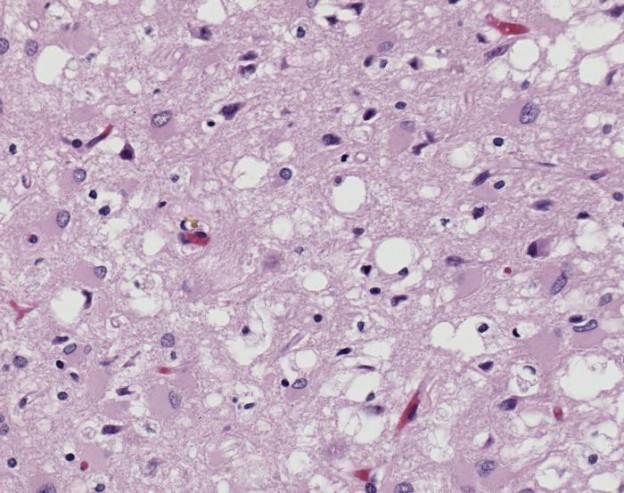
Magnified 100X, and stained with H&E (hematoxylin and eosin) staining technique, this light photomicrograph of brain tissue reveals the presence of prominent spongiotic changes in the cortex, and loss of neurons in a case of variant Creutzfeldt-Jakob disease (vCJD). Image/CDC















Thanks for sharing your thoughts about Outbreak News.
Regards
Saturday, April 6, 2013
Unique drug screening approach for prion diseases identifies tacrolimus and astemizole as antiprion agents
http://prionpps.blogspot.com/2013/04/unique-drug-screening-approach-for.html